Scroll to:
Importance of Novel Intraperitoneal Therapy of NF-kappa B Inhibitor DHMEQ
https://doi.org/10.24060/2076-3093-2019-9-1-87-88
For citations:
Gantsev Sh.Kh., Umezawa K. Importance of Novel Intraperitoneal Therapy of NF-kappa B Inhibitor DHMEQ. Creative surgery and oncology. 2019;9(1):87-88. https://doi.org/10.24060/2076-3093-2019-9-1-87-88
There have been great advances in the therapy of cancer and leukemia. But it is still true that many neoplastic diseases are difficult to treat. Especially, there is often no way to treat difficult ones such as hormone-insensitive breast carcinoma and prostate carcinoma, pancreatic carcinoma, cholangio- carcinoma, and multiple myeloma.
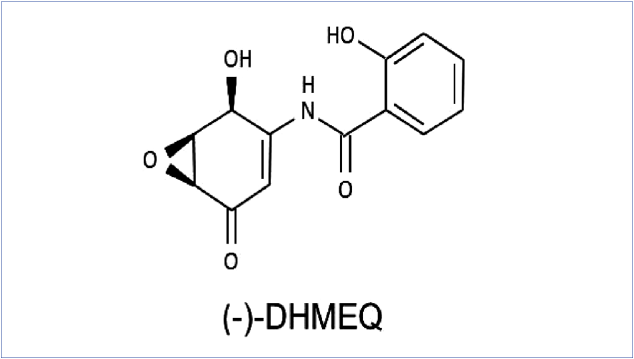
Figure 1. Structure of DHMEQ, an NF-kappa B inhibitor
NF-kappa B is a transcription factor that enhance expressions of many inflammatory cytokines. It has a role on activation of immunity and on tissue stability. But excess activation of NF-kappa B especially in macrophages often causes inflammation and cancer. Around 2000, dehydroxy- methylepoxyquinomicin (DHMEQ, fig. 1) was discovered by Umezawa as an inhibitor of NF-kappa B [1].
It was shown to be a very specific inhibitor that directly binds to and inactivates NF-kappa B components [2]. Until now DHMEQ has been used by many scientists in the world to suppress animal models of cancer and inflammation (3). Especially it suppressed difficult cancer models such as hormone-insensitive breast cancer and prostate cancer, and multiple myeloma.
All the results in figure 2 were published in various international journals including the top class ones such as Cancer Research and Blood. Moreover, no toxicity has been reported ever. Chemicals are usually injected into the peritoneal cavity in rodent experiments because of the easy technique.
Later, Umezawa suggested that intraperitoneal (IP) administration itself is important for the effectiveness and safety of DHMEQ [3-5], as shown in figure 3.
DHMEQ does not go to systemic circulation. DHMEQ inhibits inflammatory reaction in human peritoneal cells [6]. Gantsev and Umezawa began collaboration on the clinical use of DHMEQ IP therapy in Bashkortostan State Medical University in 2013. Kickoff Meeting with the President of Bashkortostan State was broadcasted in TV. PeritonTreat Ltd was founded in Ufa for the development of DHMEQ IP therapy. Since then, marked progress has been made. For example, in the occasion of BRICS Summit in Ufa in 2015, we organized Satellite Symposium of BRICS Summit entitled “Development of novel drug DHMEQ in BRICS countries”. Now we are very close to the clinical use of DHMEQ IP therapy on difficult cancer. It is now an international project including Russia, Japan, and Kazakhstan.
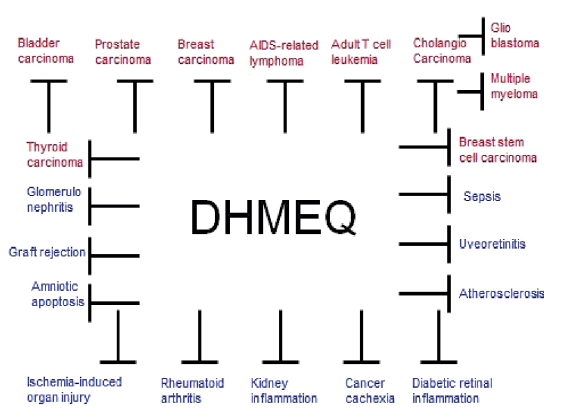
Figure 2. Suppression of various inflammatory and neoplastic disease models in animal experiments by intraperitoneal administration of DHMEQ
Anti-PD-1 antibody and anti-CTLA-4 antibody therapies are effective on melanoma and other cancers. However, thy say, anti-PD-1 therapy is effective on 20-30 % of even melanoma. The rest 70-80 % of patients are non-responding. These therapies are immune-activation therapy. Instead, DHMEQ IP therapy is an immunosuppressive therapy. It may be effective on the patients who are resistant to im- mune-activation therapy.
DHMEQ has been used in many laboratories in the world to show effectiveness to various cancer and inflammation animals. The therapy is extremely safe because of the IP therapy. If it can be used for patients, there appears a good possibility to save many patients suffering cancer and severe inflammation.
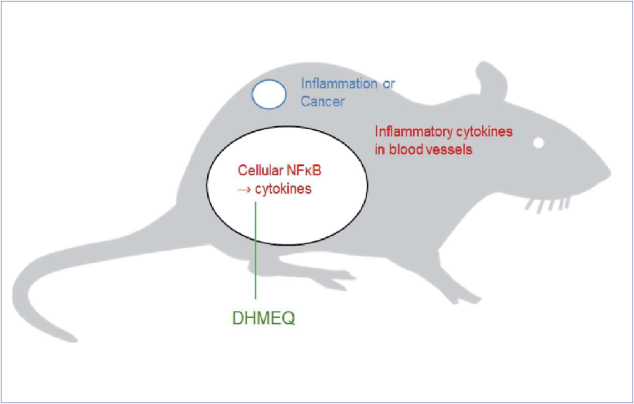
Figure 3. Target of DHMEQ is likely to be NF-kappa B of peritoneal cells. IP administration of DHMEQ lowers inflammatory cytokines in the blood vessels to inhibit local inflammation and cancer
References
1. Ariga A., Namekawa J., Matsumoto N., Inoue J., Umezawa K. Inhibition of tumor necrosis factor-alpha -induced nuclear translocation and activation of NF-kappa B by dehydroxymethylepoxyquinomicin. J Biol Chem. 2002;277(27):24625–30. DOI: 10.1074/jbc.M112063200
2. Yamamoto M., Horie R., Takeiri M., Kozawa I., Umezawa K. Inactivation of nuclear factor kappa B components by covalent binding of (−)-dehydroxymethylepoxyquinomicin to specific cysteine residues. J Med Chem. 2008;51(18):5780–8. DOI: 10.1021/jm8006245
3. Lin Y., Ukaji T., Koide N., Umezawa K. Inhibition of late and early phases of cancer metastasis by NF-κB inhibitor DHMEQ derived from microbial bioactive metabolite epoxyquinomicin: A review. Int J Mol Sci. 2018;19(3):729. DOI: 10.3390/ijms19030729
4. Umezawa K. Possible role of peritoneal NF-κB in peripheral inflammation and cancer: Lessons from the inhibitor DHMEQ. Biomed Pharmacother. 2011;65(4):252–9. DOI: 10.1016/j.biopha.2011.02.003
5. Umezawa K. Peritoneal NF-κB as a possible molecular target for suppression of various cancers and inflammation. (Review) Forum Immunopathol Dis Therap. 2013;4(1):63–77. DOI: 10.1615/ForumImmunDisTher.2013008314
6. Sosinnska P., Macckowiak B., Staniszewski R., Umezawa K., Breborowicz A. Inhibition of NF-κB with dehydroxyepoxiquinomicin modifies function of human peritoneal mesothelial cells. Am J Transl Res. 2016;8(12):5756–5. PMID: 28078047
About the Authors
Sh. Kh. GantsevRussian Federation
Gantsev Shamil — Doctor of Medical Sciences, Professor, Head of the Department of Oncology with Oncology and Anatomical Pathology courses in the Institute of Additional Professional Education
3 Lenin str., Ufa, 450008
Kazuo Umezawa
Japan
Umezawa Kazuo — Professor of the Department of Molecular Target Medicine Screening
Review
For citations:
Gantsev Sh.Kh., Umezawa K. Importance of Novel Intraperitoneal Therapy of NF-kappa B Inhibitor DHMEQ. Creative surgery and oncology. 2019;9(1):87-88. https://doi.org/10.24060/2076-3093-2019-9-1-87-88


































